A Pregnant Woman with Fever and Respiratory Failure
Reviewed By Microbiology, Tuberculosis & Pulmonary Infections Assembly
Submitted by
Sonia Vishin, MD
Fellow
Division of Pulmonary, Allergy and Critical Care Medicine
University of Alabama at Birmingham
Jody Hunt, MD
Fellow
Division of Pulmonary, Allergy and Critical Care Medicine
University of Alabama at Birmingham
Keith Wille, MD
Associate Professor
Division of Pulmonary, Allergy and Critical Care Medicine
University of Alabama at Birmingham
Submit your comments to the author(s).
History
A 28-year-old white female at 29 weeks gestation, G2P1, presented to the emergency department with increasing shortness of breath and fever for 3 days. She also endorsed nausea, loose bowel movements and emesis for 2 days prior to admission. Prior to this, she was in her usual state of health. She denied cough, chest pain, joint pain, rash or hemoptysis. Her past medical history was significant for well-controlled asthma. However, over the last few days, she had been using her rescue inhaler up to 8 times a day. She denied any sick contacts. She had one previous full-term pregnancy and delivered vaginally, with no complications. Her family history was significant for both parents having hypertension and diabetes mellitus. Her father also had congestive heart failure. The patient smokes one pack of cigarettes per day but denies alcohol or illicit drug use. She is married and lives with her husband and child. Her medications at the time of admission included folic acid, prenatal vitamins, fluticasone/salmeterol and albuterol MDI as needed.
Physical Exam
On examination, the patient’s pulse was 94 beats per minute and regular. She had a temperature of 101.3 degrees Fahrenheit and a blood pressure of 98/31 mm Hg. Her oxygen saturation was 84% on 100% O2 by nonrebreather mask and had a respiratory rate of 32 breaths per minute. Cardiovascular exam revealed no murmurs. Respiratory exam showed bilateral crackles. Abdominal exam showed gravid uterus, otherwise unrevealing.
Lab
- Serum chemistries were significant for a bicarbonate level of 18 mEq/L and creatinine of 1.5 mg/dl
- Complete blood count was significant for WBC 12.5 x 103, platelet count 145 x 103, hemoglobin 9.4 g/dl. Differential count was 90% segmented neutrophils and 8% lymphocytes
- Urinalysis showed 3+ blood, 1+ ketones, 1+ protein
- Liver function profile was significant for AST 148, ALT 32
- Rapid flu test was negative
Chest radiograph is shown in Figure 1.
She was intubated urgently for respiratory failure. Based on the chest radiograph and lab studies, further work up was done, including bronchoscopy with bronchoalveolar lavage (BAL):
- BAL fluid:
- CMV culture negative
- Viral culture negative
- Viral respiratory panel positive for Influenza A
- Legionella culture negative
- Blood cultures negative
- Urine Legionella antigen negative
- Transthoracic echocardiogram – bilateral ejection fractions >55%, no vegetations, no significant valvular disease
Figures
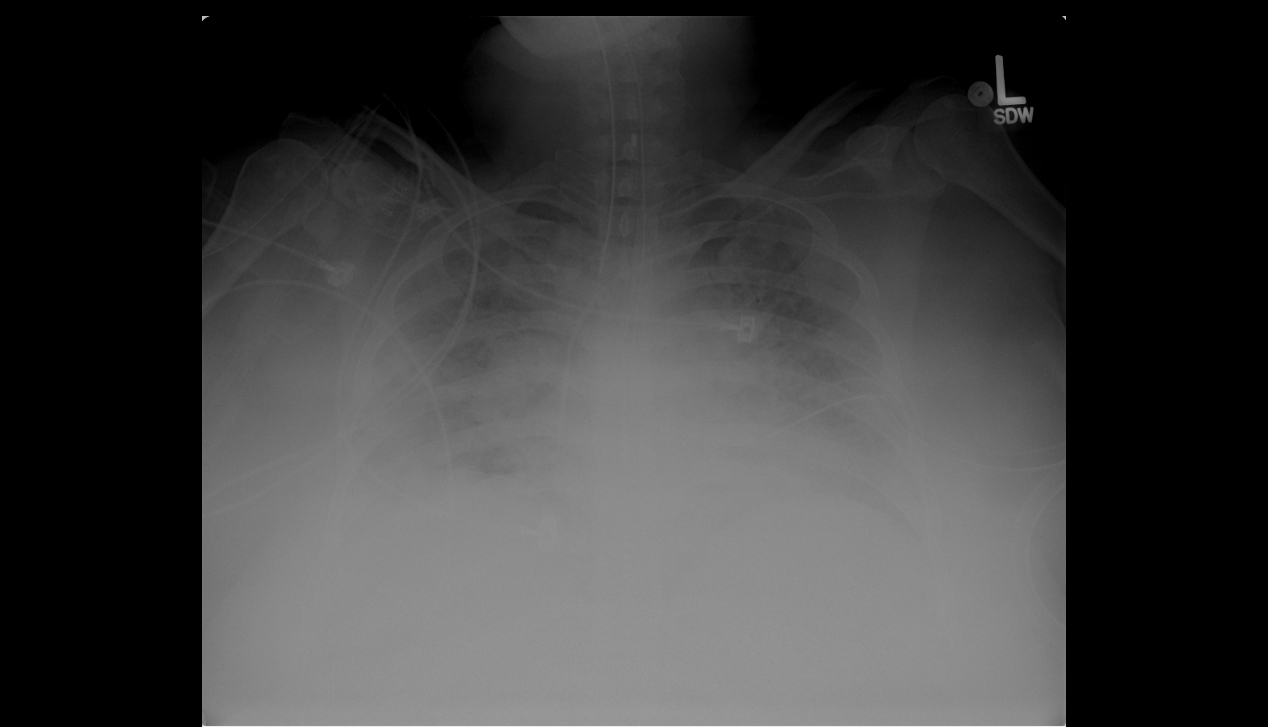
Figure 1. Portable chest radiograph
Influenza A (H1N1) is a novel strain which has reached pandemic levels this year. At the time of her illness, 97% of the circulating influenza A virus was the novel strain, H1N1(1). The virus is spread by respiratory droplets and by contact with contaminated surfaces. The groups at highest risk for influenza A (H1N1) include pregnant women, children <5 years of age, the elderly, people with chronic co-morbid conditions such as liver disease or heart disease and patients who are immunosuppressed. The clinical presentation is similar to seasonal Influenza and includes high fevers, cough, shortness of breath, rhinorrhea, headache, myalgias, nausea and vomiting (3). Rapid influenza testing is reported to have a low sensitivity (10-50%) for novel influenza A (H1N1), and therefore rapid testing methods should not be relied upon to exclude the presence of disease. The diagnosis is established definitively by PCR analysis of respiratory fluid (3). However, the diagnosis is highly suspected in patients with positive rapid testing for influenza A and treatment should be initiated immediately. Pregnant women are at higher risk during their pregnancy and 2 weeks post-partum(4). Poorer outcomes in mothers and newborns have been associated with novel influenza A. The spectrum of perinatal complications includes: preterm labor and birth, pneumonia, acute respiratory distress syndrome and death. Some risk factors among pregnant patients have been identified and include obesity, current cigarette smoking and third trimester of gestation (4).
Further history
This patient ultimately had BAL fluid PCR test positive for influenza A H1N1. She developed ARDS and had a prolonged ICU course on mechanical ventilatory support.
There are two major options for treatment of influenza A (H1N1), oseltamivir and zanamivir, both of which are neuraminidase inhibitors. These drugs act by preventing further spread of virus within the respiratory tract. Initiation of therapy should not wait for confirmatory diagnostic testing. The dosing of oseltamivir is 75mg orally twice daily for a minimum of 5 days (2). The optimal length of treatment in critically ill patients is not known. The current recommendations state only that persons with severe infections, such as those who require ICU admission or with prolonged infection, may require longer treatment courses (1). It has also been suggested that double strength oseltamivir, 150mg orally twice a day, may be used for critically ill patients, although there is limited data to support this (2). It is recommended that treatment begin within 48-72 hours of onset of symptoms, although patients may still benefit from starting antiviral medications after that period (5). Although both oseltamivir and zanamivir are approved for use in pregnancy, the Center for Disease Control (CDC) guidelines currently recommend using oseltamivir as first line therapy (4). Both medications are pregnancy category C. Peramivir is a new neuraminidase inhibitor that is yet to be approved for use by the FDA, as it is currently in Phase 3 clinical trials. However, given the public health emergency of influenza A H1N1, it has received emergency approval for use in patients who are refractory to the usual neuraminidase inhibitors or to those who are unable to take oral medications. It is not first line therapy. Oseltamivir-resistant virus has been reported in immunocompromised hospitalized patients in 2 outbreaks. Transmission of resistant virus from one patient to another is suspected in both outbreaks. All of the resistant viruses carried the same H275Y mutation, indicating resistance to oseltamivir but susceptibility to the second antiviral drug, zanamivir (6). It is recommended to consider switching to zanamivir in an immunocompromised, hospitalized patient with ongoing illness despite oseltamivir administration. Zanamivir should be considered first line treatment in hospital wards where oseltamivir resistance has been discovered. (5)
Further history
After two weeks of ICU care, the patient underwent a tracheostomy. Her hospitalization was prolonged, but gradually she improved, was weaned from ventilatory support and discharged. The effects of this illness on her pregnancy and fetus are as yet unknown.
The CDC recommends that the first groups who should be immunized against novel influenza A (H1N1) include pregnant women, caretakers of children under the age of 6 months, healthcare and emergency service workers, those between the ages of 6 months and 24 years and those aged 24-64 years who have chronic co-morbid conditions that put them at higher risk (1). Once all these at-risk groups receive the vaccine, it will then be offered to all persons above the age of 65. The FDA has approved a one-dose vaccine for all persons above the age of 10. Infants under the age of 6 months should not be immunized. There are two vaccine forms available. The first is an inactivated virus vaccine, which is administered by intramuscular injection. The second is a live virus vaccine, which is administered intranasally. The CDC and FDA are monitoring adverse side effects of the vaccine through the Vaccine Adverse Events Reporting System (VAERS) which is a national reporting site. As of November 20, 2009 the adverse event profile has been similar to that of the seasonal influenza vaccine (7).
References
- Updated interim recommendations for the use of antiviral medications in the treatment and prevention of influenza for the 2009-2010 flu season. Centers for Disease Control Website. Available from: http://www.cdc.gov/H1N1flu/recommendations.htm.
- Treanor JJ, Hayden FG, Vrooman PS, et al. Efficacy and safety of the oral neuraminidase inhibitor oseltamivir in treating acute influenza. JAMA 2000;282:1016-1024.
- Harper SA, Bradley JS, Englund JA, et al. Seasonal influenza in adults and children-diagnosis, treatment, chemoprophylaxis, and institutional outbreak management: clinical practice guidelines of the Infectious Diseases Society of America. Clin Infect Dis 2009;48:1003-1032.
- Updated Interim Recommendations for Obstetric Health Care Providers Related to Use of Antiviral Medications in the Treatment and Prevention of Influenza for the 2009-2010 Season; Center for Disease Control Website. Available from: http://www.cdc.gov/H1N1flu/pregnancy/antiviral_messages.htm.
- World Health Organization, Global Alert and Response; Oseltamivir Resistance in Immunocompromised Hospitalized Patients. Available from: http://www.who.int/csr/disease/swineflu/notes/briefing_20091202/en/index.html.
- Hurt AC, Holien JK, Parker MW, et al. Oseltamivir Resistance and the H274Y Neuraminidase Mutation in Seasonal, Pandemic and Highly Pathogenic Influenza Viruses. Drugs 2009;69:2523-2531.
- Summary of 2009 Monovalent H1N1 Influenza Vaccine Data- Vaccine Adverse Event Reporting System; Centers for Disease Control. Available from: http://vaers.hhs.gov/resources/2009H1N1Summary_Nov25.pdf.
- Sym D, Patel PN, El-Chaar GM. Seasonal, avian, and novel H1N1 influenza: Prevention and treatment modalities. Ann Pharmacother 2009;43:2001-2011.
- Jamieson DJ, Honein MA, Rasmussen SA, et al. H1N1 2009 influenza virus infection during pregnancy in the USA. Lancet 2009;374:451-458.
- Saleeby E, Chapman J, Morse J, et al. H1N1 influenza in pregnancy: cause for concern. Obstet Gynecol 2009;114:885-891.




